|
Three types of moderators are used at the MIT reactor: (1) ordinary or "light" water that is also used to cool the reactor core, (2) deuterated or heavy water (D 20), and (3) high-purity graphite, both of which are excellent at slowing neutrons without absorbing them. Since U-235 nuclei do not readily absorb the high energy neutrons that are emitted during fission, it is necessary to slow the neutrons down with a "moderator". In the MIT reactor, one other group of components is essential to the maintaining and controlling a chain reaction. The balanced nuclear equation for the fission reaction n 0 1 + U 92 235 K 36 91 r + B 56. Most absorption reactions result in fission reaction, but a part of reactions result in radiative capture forming 240 Pu. Write the balanced nuclear equation for the fission reaction. For fast neutrons, its fission cross-section is on the order of barns. A nuclear equation is an equation that describes the reactants and products in nuclear fission, nuclear fusion, or one of the four types of radioactive decay. To maintain the steady rate of fission, the neutron absorbing material. As fewer and fewer neutrons are absorbed, more and more neutrons are available to cause the splitting of uranium nuclei, until finally enough neutrons are available to sustain a chain reaction. Plutonium 239 is a fissile isotope, and its fission cross-section for thermal neutrons is about 750 barns (for 0.025 eV neutron). U235 + 0n1 56Ba141 + 36Kr92 + 3(0n1) +200MeV. To put the reactor into operation, the control blades are raised very slowly. During nuclear fission, a neutron collides with a uranium atom and splits it, releasing a large amount of energy in the form of heat and radiation. Just as we use the number and type of atoms present to balance a chemical equation, we can use the number and type of nucleons present to write a balanced nuclear equation for a nuclear decay reaction. When the control blades are fully inserted, they absorb so many neutrons from the uranium that there are not enough to allow a chain reaction to continue. Boron has the property of absorbing neutrons without re-emitting any. Example One Calculate the amount of energy released for the fission reaction of the 235 U when 144 Cs and 90 Rb are two neutrons. The balanced nuclear equation for the reaction is as follows: 3015P 3014Si + 0+1 15 30 P 14 30 Si + + 1 0. The rate of fissions in the uranium nuclei in the MIT reactor is controlled chiefly by six control blades of boron-stainless steel which are inserted vertically alongside the fuel elements. Write the nuclear equation for the beta decay of cesium-137. In this section, we are going to solve two examples of the Nuclear Fission Equation to clarify this topic more. B The mass number of the second product is A 30 0 30, and its atomic number is Z 15 1 14, which corresponds to silicon. Definition Definition Symbolic representation of a nuclear reaction which uses symbols to represent the protons, neutrons, alpha particles, beta particles, positrons, and gamma rays involved. Assume the nuclei shown represent 235 U splitting into 92 Kr and 141 Ba. When it is in operation, the central active core contains a huge number of neutrons traveling in every direction at very high speeds. Write a balanced nuclear equation to represent the image above that depicts nuclear fission. The MIT Research Reactor is used primarily for the production of neutrons. Hence, the possibility exists for creating a chain reaction. Each time a U-235 nucleus splits, it releases two or three neutrons. This process is known as fission (see diagram below). When a U-235 nucleus absorbs an extra neutron, it quickly breaks into two parts. The arrangement of particles within uranium-235 is somewhat unstable and the nucleus can disintegrate if it is excited by an outside source. For a proton or a neutron, A = 1.In the nucleus of each atom of uranium-235 (U-235) are 92 protons and 143 neutrons, for a total of 235.  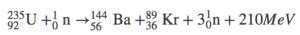 
Thus the sum of the mass numbers of the products (118 + 132 + 4 254. Nuclei consist of nucleons (neutrons and protons), the total number of which is equal to the mass number of the nucleus. Once again, the number of nucleons is conserved. Nuclear fission - Atomic Reactions, Energy Release, Chain Reactions: The fission process may be best understood through a consideration of the structure and stability of nuclear matter. Like the notation used to indicate isotopes, the upper left superscript in the symbol for a particle gives the mass number, which is the total number of protons and neutrons. An example is the spontaneous fission of 98 254 Cf, which gives a distribution of fission products one possible set of products is shown in the following equation: (19.5.1) 98 254 Cf 46 118 Pd + 52 132 Te + 4 0 1 n. \): Nuclear Decay Emissions and Their Symbols Identity
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
